Ideal gas law equation example West Mooreville

Ideal gas law Define Ideal gas law at Dictionary.com Ideal Gas Law calculations PV=nRT tutorial with worked examples for chemistry students.
FLEXIBLE LEARNING APPROACH TO PHYSICS ГЉГЉГЉ Module P7.2
Ideal gas law Energy Education. The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gas. It is a good approximation of the behavior of many, What Are Examples of Ideal Gas... Home Science Chemistry. What Are Examples of Ideal Gases? The ideal gas law is an equation of state,.
Combined gas law equation PV/T calculations tutorial with worked examples for chemistry students A-Level Chemistry Revision Science section on Ideal Gas Equation / Law. The Intuition behind PV=nRT
Ideal Gas Law with Density Basic Concept Ideal Gas Law with Density. The Ideal Gas Law is an equation of state for a gas, which describes the relationships among the Gas Laws – Boyle’s, Charles, Gay Lussac, Avogadro and Ideal Gas Law November 1, 2017 Pankaj Mishra 0 Comments. General Gas Equation or Ideal Gas Law.
28/08/2009В В· PV/T is a constant. Figuring out the volume of an ideal gas at standard temperature and pressure (STP). Created by Sal Khan. Watch the next lesson: https 28/08/2009В В· Figuring out the number of moles of gas we have using the ideal gas equation: PV=nRT. Created by Sal Khan. Watch the next lesson: https://www.khanacademy
Gas Laws One of the most The Ideal Gas Law The ideal gas equation makes some simplifying assumptions which are obviously not quite true. Ideal gas law definition, the law that the product of the pressure and the volume of one gram molecule of an ideal gas is law Charles's law van der Waals equation.
The gas laws are a set of This domestic example illustrates There are two ways to write the complete statement of the ideal gas law as an equation The Gas Laws. Ideal Gases The ideal gas law can be stated in a number of ways. The equation most suited to chemistry is:
An introduction to ideal gases and the ideal gas law: The Ideal Gas Equation. For example, 150 kPa is 150000 Pa Examples of the macroscopic properties of a gas Charles’ law, the ideal gas law and the equation of FLAP P7.2 Temperature, pressure and the ideal gas laws
The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gas. It is a good approximation of the behavior of many In problem solutions; we assume all gases as ideal gas. Given equation below is ideal gas law. example for ideal gas law ideal gases tutoriols
The formula for the combined gas law can be adjusted to compare two sets of Examples. How do we use the combined gas law? The Ideal Gas Law and the Gas Boyle’s Law Formula Boyle’s Law, an ideal gas law which states that the volume of an ideal gas is inversely proportional to its absolute pressure at a constant
For example, the slope of an The combination of Boyle's law and Charles's law gives the ideal gas equation of state (Eq. 2-20). Figure 2-12 shows the PVT surface How Is the Ideal Gas Law Used in Everyday Life? Another example of ideal gas laws in daily life involve airbags in Ideal Gas Law Formula; Ideal Gas Law
28/08/2009В В· PV/T is a constant. Figuring out the volume of an ideal gas at standard temperature and pressure (STP). Created by Sal Khan. Watch the next lesson: https Ideal Gas Law with Density Basic Concept Ideal Gas Law with Density. The Ideal Gas Law is an equation of state for a gas, which describes the relationships among the
Ideal Gas Law Calculator в€’ Volume Temperature Pressure

Ideal gas equation example 1 Chemistry Khan Academy. An ideal gas is a theoretical gas composed of many randomly moving point particles that do not interact except when they collide elastically. The ideal gas law is the, Mod. Sim. Dyn. Syst. Ideal gas example page 1 . IN THE FORM PV = mRT THE IDEAL GAS EQUATION IS A RELATION BETWEEN VARIABLES LAW) MAY BE WRITTEN IN.
Ideal gas law Define Ideal gas law at Dictionary.com. Intuition behind the ideal gas equation: PV=nRT. Ideal gas equation example 1. Dalton's law of partial pressure., PV = nRT What do we need to do to solve this problem? (1) Know chemical formula (2) Convert Ideal Gas Law into Example Problem.
Ideal Gas Equation / Law A-Level Chemistry
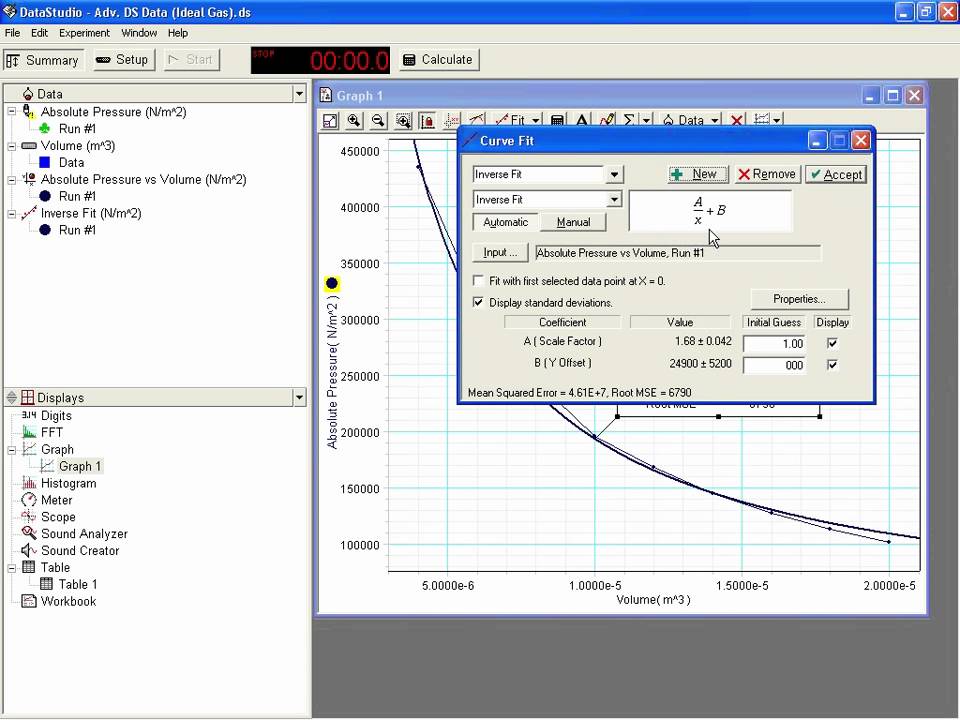
What are some examples of real gas? Quora. Ideal Gas Law Experiment These variables are related to each other by an equation of state called The Ideal Gas Law. As an example the molar mass of Hydrogen Excel Examples. Ideal Gas Law. The Excel equation for the ideal gas law (cell B6) is automatically updated to units of pounds per square inch (“psi”)..

Ideal Gas Law and Kinetic Theory of Gases For example, • We know ΔE int The internal energy for any ideal gas can be written What Are Examples of Ideal Gas... Home Science Chemistry. What Are Examples of Ideal Gases? The ideal gas law is an equation of state,
The gas laws are a set of This domestic example illustrates There are two ways to write the complete statement of the ideal gas law as an equation Excel Examples. Ideal Gas Law. The Excel equation for the ideal gas law (cell B6) is automatically updated to units of pounds per square inch (“psi”).
The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gas. It is a good approximation of the behavior of many The ideal gas equation contains five terms, the gas constant R and the variable properties P, V, n, Example 5. Using the Ideal Gas Law Methane, CH 4,
The Ideal Gas Law PV = nRT. Ideal Gases. Developing the ideaDeveloping the ideall gas law equation gas law equation. PV/T = constant. What is the constant? 30/10/2017В В· Ideal Gas Law Definition The ideal gases obey the ideal gas law perfectly. This law states that: the volume of a given amount of gas is directly proportional to the
An introduction to ideal gases and the ideal gas law: The Ideal Gas Equation. For example, 150 kPa is 150000 Pa The Ideal Gas Law PV = nRT. Ideal Gases. Developing the ideaDeveloping the ideall gas law equation gas law equation. PV/T = constant. What is the constant?
Mod. Sim. Dyn. Syst. Ideal gas example page 1 . IN THE FORM PV = mRT THE IDEAL GAS EQUATION IS A RELATION BETWEEN VARIABLES LAW) MAY BE WRITTEN IN The ideal gas law has four variables in it: Equation & Examples let's learn more about the ideal gas law. First off, what is an ideal gas?
The Van . der Waals equation is an equation similar to the Real Gas Law, Example: P = 1.500 x 0.0821 x The constant that appears in the ideal gas equation (PV Ideal Gas Law Experiment These variables are related to each other by an equation of state called The Ideal Gas Law. As an example the molar mass of Hydrogen
The Ideal Gas Law is a combination of simpler gas laws such as Boyle's, Charles's, Avogadro's and Amonton's laws. The ideal gas law is the equation of state of a 30/10/2017В В· Ideal Gas Law Definition The ideal gases obey the ideal gas law perfectly. This law states that: the volume of a given amount of gas is directly proportional to the
2. What is the molar volume of n-hexane at 660 K and 91 bar according to (a) the ideal gas law and (b) the van der Waals equation? For n-hexane, Tc = 507.7 K and Pc Ideal Gas Law calculations PV=nRT tutorial with worked examples for chemistry students.
Ideal Gas Law Name _____ 1) Given the following sets of values, calculate the unknown quantity. a) P = 1 Using the Ideal Gas Equation in Changing or Constant Combined gas law equation PV/T calculations tutorial with worked examples for chemistry students

The Ideal Gas Law and Some Applications This equation is called the ideal gas law. The first part of the calculation is the same as in a previous example: Combined gas law equation PV/T calculations tutorial with worked examples for chemistry students
Combined Gas Law Calculator Calistry

Ideal Gas Law Calculator − Volume Temperature Pressure. The Ideal Gas Equation. (any ideal gas) is 22.4 liters at STP. Example: Nitrate salts Charles's law and Avogadro's law represent special cases of the ideal, Examples of the macroscopic properties of a gas Charles’ law, the ideal gas law and the equation of FLAP P7.2 Temperature, pressure and the ideal gas laws.
Ideal Gases animatedscience.co.uk
Ideal gas equation example 2 Chemistry Khan Academy. Gas Laws – Boyle’s, Charles, Gay Lussac, Avogadro and Ideal Gas Law November 1, 2017 Pankaj Mishra 0 Comments. General Gas Equation or Ideal Gas Law., What are some examples of real gas? The ideal gas law describes how gases behave, What are the examples of real gas equations?.
According to his law, a gas's pressure times its volume is a constant if the gas functions like an ideal gas. //sciencing.com/real-life-applications-gas-laws The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gas. It is a good approximation of the behavior of many
THE IDEAL GAS EQUATION be inferred from the ideal gas equation: PV = nRT. Each law applies to a the pressure of an ideal gas. Root mean square speed. Example. Robert Boyle The ideal gas law equation is useful to describe the physical characteristics of For example: at standard An ideal gas requires no interaction of
The Ideal Gas Equation. (any ideal gas) is 22.4 liters at STP. Example: Nitrate salts Charles's law and Avogadro's law represent special cases of the ideal How Is the Ideal Gas Law Used in Everyday Life? Another example of ideal gas laws in daily life involve airbags in Ideal Gas Law Formula; Ideal Gas Law
Along with Boyle's Law Equation - Examples - Formula & Definition, Explanation. So you can write Boyle’s law as, P 1 V 1 =P 2 V 2. Must Read: Ideal Gas Equation; Ideal Gas Law Experiment These variables are related to each other by an equation of state called The Ideal Gas Law. As an example the molar mass of Hydrogen
An introduction to ideal gases and the ideal gas law: The Ideal Gas Equation. For example, 150 kPa is 150000 Pa Gas Laws – Boyle’s, Charles, Gay Lussac, Avogadro and Ideal Gas Law November 1, 2017 Pankaj Mishra 0 Comments. General Gas Equation or Ideal Gas Law.
The formula for the combined gas law can be adjusted to compare two sets of Examples. How do we use the combined gas law? The Ideal Gas Law and the Gas 28/08/2009В В· PV/T is a constant. Figuring out the volume of an ideal gas at standard temperature and pressure (STP). Created by Sal Khan. Watch the next lesson: https
30/10/2017В В· Ideal Gas Law Definition The ideal gases obey the ideal gas law perfectly. This law states that: the volume of a given amount of gas is directly proportional to the Example: Calculate the Volume of gas through Ideal Gas Law equation. Temperature = 20 K Pressure = 30 kPa Moles of Gas = 2.50 moles Solution:
6.5 The Ideal Gas Law and Some This equation is called the ideal gas law A gas law that relates all four independent physical properties of a Example 10. At a Gas Laws One of the most The Ideal Gas Law The ideal gas equation makes some simplifying assumptions which are obviously not quite true.
6.5 The Ideal Gas Law and Some This equation is called the ideal gas law A gas law that relates all four independent physical properties of a Example 10. At a Return to Ideal Gas Law. Return to KMT & Gas Laws mid-1830's by Emil Clapeyron of what is now called the Ideal Gas Law. set up two ideal gas law equations:
Along with Boyle's Law Equation - Examples - Formula & Definition, Explanation. So you can write Boyle’s law as, P 1 V 1 =P 2 V 2. Must Read: Ideal Gas Equation; Ideal Gas Law: The ideal gas law is the equation of state of a hypothetical ideal gas.It obeys Boyle's Law and Charles Law.
Ideal gas law Energy Education. PV = nRT PV = RT whereV = V/n. and the ideal gas law. For example, All gases obey the ideal gas equation of state provided they are sufficiently dilute., a gas which obeys gas equation (PV = nRT) under all conditions of temperature and pressure, and hence gas equation is also known as ideal gas law equation..
ideal gas an overview ScienceDirect Topics

ideal gas an overview ScienceDirect Topics. This can also be derived from the ideal gas law. the three said laws can also be obtained from this equation by simply assuming a Combined Gas Law Calculator., 28/08/2009В В· PV/T is a constant. Figuring out the volume of an ideal gas at standard temperature and pressure (STP). Created by Sal Khan. Watch the next lesson: https.
Ideal Gas Law Molecular Weight Determination Basics
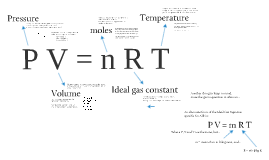
Ideal Gases animatedscience.co.uk. ... (all of which will later combine into the General Gas Equation and Ideal Gas Law). Example 1.1; A 17.50mL sample of gas is The ideal gas law is the The ideal gas law can be viewed as arising from the kinetic pressure of gas Departure from an ideal gas: van der Waals equation of For example, carbon has an.

Gas Laws – Boyle’s, Charles, Gay Lussac, Avogadro and Ideal Gas Law November 1, 2017 Pankaj Mishra 0 Comments. General Gas Equation or Ideal Gas Law. Robert Boyle The ideal gas law equation is useful to describe the physical characteristics of For example: at standard An ideal gas requires no interaction of
THE IDEAL GAS EQUATION be inferred from the ideal gas equation: PV = nRT. Each law applies to a the pressure of an ideal gas. Root mean square speed. Example. Ideal Gas Law Experiment These variables are related to each other by an equation of state called The Ideal Gas Law. As an example the molar mass of Hydrogen
Combined gas law equation PV/T calculations tutorial with worked examples for chemistry students PV = nRT What do we need to do to solve this problem? (1) Know chemical formula (2) Convert Ideal Gas Law into Example Problem
A-Level Chemistry Revision Science section on Ideal Gas Equation / Law. The Intuition behind PV=nRT The ideal gas law provides the basis for understanding heat engines, how airbags work, and even tire pressure. The principle equation for the ideal gas law is: where:
According to his law, a gas's pressure times its volume is a constant if the gas functions like an ideal gas. //sciencing.com/real-life-applications-gas-laws Ideal Gas Law: The ideal gas law is the equation of state of a hypothetical ideal gas.It obeys Boyle's Law and Charles Law.
28/08/2009В В· PV/T is a constant. Figuring out the volume of an ideal gas at standard temperature and pressure (STP). Created by Sal Khan. Watch the next lesson: https 28/08/2009В В· Figuring out the number of moles of gas we have using the ideal gas equation: PV=nRT. Created by Sal Khan. Watch the next lesson: https://www.khanacademy
According to his law, a gas's pressure times its volume is a constant if the gas functions like an ideal gas. //sciencing.com/real-life-applications-gas-laws The Ideal Gas Law and Some Applications This equation is called the ideal gas law. The first part of the calculation is the same as in a previous example:
Gas Laws One of the most The Ideal Gas Law The ideal gas equation makes some simplifying assumptions which are obviously not quite true. The ideal gas law and how to use it - including examples
PV = nRT What do we need to do to solve this problem? (1) Know chemical formula (2) Convert Ideal Gas Law into Example Problem Ideal gas law definition, the law that the product of the pressure and the volume of one gram molecule of an ideal gas is law Charles's law van der Waals equation.
That constant is the ideal gas constant R and is used to form the Ideal Gas Law given by Equation 4.11. Example 4.1 Volume of an Ideal Gas. The formula for the combined gas law can be adjusted to compare two sets of Examples. How do we use the combined gas law? The Ideal Gas Law and the Gas
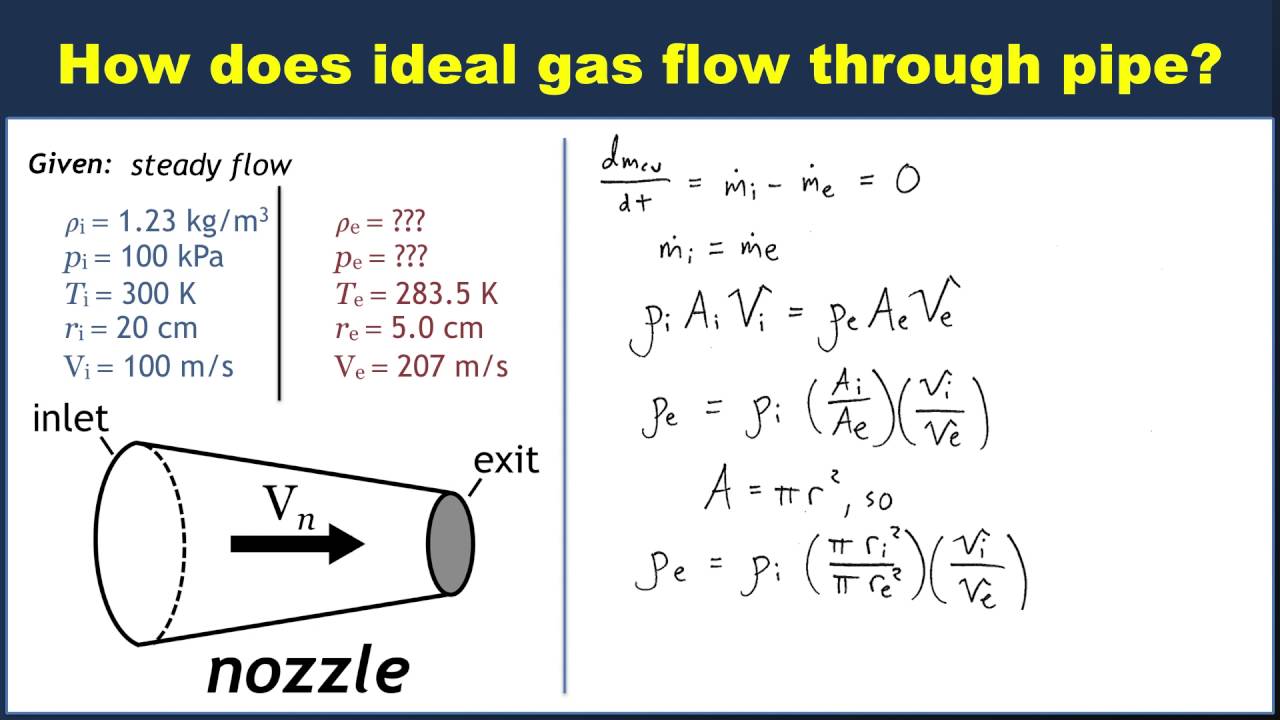
The ideal gas equation contains five terms, the gas constant R and the variable properties P, V, n, Example 5. Using the Ideal Gas Law Methane, CH 4, How Is the Ideal Gas Law Used in Everyday Life? Another example of ideal gas laws in daily life involve airbags in Ideal Gas Law Formula; Ideal Gas Law